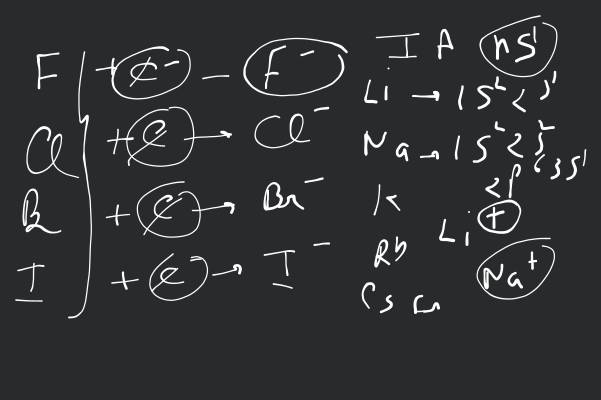Question
Question asked by Filo student

The alkali metal halides, are all high melting, colourless crystalline solids. They can be prepared by the reaction of the appropriate oxide, hydroxide or carbonate with aqueous hydrobalic acid (HX). All of these halides have high negative enthalpies of formation: the values for fluorides become less negative as we go down the group. whilst the reverse is true for for chlorides, bromides and iodides. For a given metal always becomes less negative from fluoride to iodide. The melting and boiling points always follow the trend: fluoride chloride bromide > iodide. All these halides are soluble in water. The low solubility of in water is due to its high lattice enthalpy whereas the low solubility of CsI is due to smaller hydration enthalpy of its two ions. Other halides of lithium are soluble in ethanol, acetone and ethylacetate: is soluble in pyridine also.
Found 4 tutors discussing this question
Discuss this question LIVE
15 mins ago
Filo tutor solutions (2)
Learn from their 1-to-1 discussion with Filo tutors.

One destination to cover all your homework and assignment needs
Learn Practice Revision Succeed

Instant 1:1 help, 24x7
60, 000+ Expert tutors

Textbook solutions
Big idea maths, McGraw-Hill Education etc

Essay review
Get expert feedback on your essay

Schedule classes
High dosage tutoring from Dedicated 3 experts
Practice more questions on s-Block
Question 1
Medium
Views: 5,216
Question 2
Easy
Views: 6,038
Question 3
Views: 5,220
Question 4
Medium
Views: 5,449
Students who ask this question also asked
Question 1
Views: 5,174
Question 4
Views: 5,440


Stuck on the question or explanation?
Connect with our Chemistry tutors online and get step by step solution of this question.
231 students are taking LIVE classes
| Question Text | The alkali metal halides, are all high melting, colourless crystalline solids. They can be prepared by the reaction of the appropriate oxide, hydroxide or carbonate with aqueous hydrobalic acid (HX). All of these halides have high negative enthalpies of formation: the values for fluorides become less negative as we go down the group. whilst the reverse is true for for chlorides, bromides and iodides. For a given metal always becomes less negative from fluoride to iodide.
The melting and boiling points always follow the trend: fluoride chloride bromide > iodide. All these halides are soluble in water. The low solubility of in water is due to its high lattice enthalpy whereas the low solubility of CsI is due to smaller hydration enthalpy of its two ions. Other halides of lithium are soluble in ethanol, acetone and ethylacetate: is soluble in pyridine also.
|
| Updated On | Mar 25, 2023 |
| Topic | s-Block |
| Subject | Chemistry |
| Class | Class 12 |
| Answer Type | Video solution: 2 |
| Upvotes | 140 |
| Avg. Video Duration | 7 min |