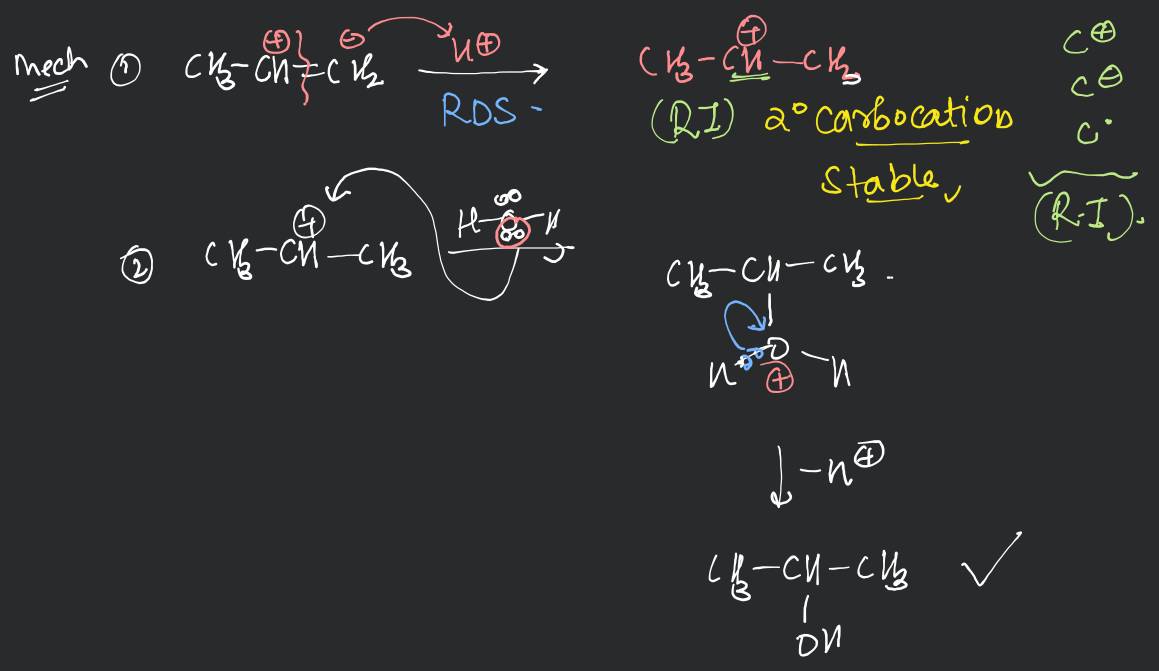Question
Question asked by Filo student
and 11.4.1 Preparation of Alcohols
Alcohols are prepared by the following methods:
1. From alkenes
(i) By acid catalysed hydration: Alkenes react with water in the presence of acid as catalyst to form alcohols. In case of unsymmetrical alkenes, the addition reaction takes place in accordance with Markovnikov's rule (Unit 13, Class XI).
Mechanism
The mechanism of the reaction involves the following three steps:
Step 1: Protonation of alkene to form carbocation by electrophilic attack of .
Step 2: Nucleophilic attack of water on carbocation.
C[C+](C)C(C)C C#CC(C)C(C)(C)[OH2+]
Step 3: Deprotonation to form an alcohol.
329 Alcohols, Phenols and Ether

Found 3 tutors discussing this question
Discuss this question LIVE
14 mins ago

One destination to cover all your homework and assignment needs
Learn Practice Revision Succeed

Instant 1:1 help, 24x7
60, 000+ Expert tutors

Textbook solutions
Big idea maths, McGraw-Hill Education etc

Essay review
Get expert feedback on your essay

Schedule classes
High dosage tutoring from Dedicated 3 experts
Practice more questions on Alcohols, Phenols & Ethers
Question 2
Medium
Views: 5,198
Question 3
Medium
Views: 6,120
(a) Enantiomers
(b) Diastereomers
(c) Meso compound
(d) Racemic modification
(E) stereoisomers that are not related as object and
(p) An optically inactive compound having plane of symmetry
(q) An equimolar mixture of enantiomers
(r) separation of a mixture of enantiomers
(s) stereoisomers that are not related as object and mirror image
Students who ask this question also asked
Question 1
Views: 5,815
Question 3
Views: 5,941


Stuck on the question or explanation?
Connect with our Chemistry tutors online and get step by step solution of this question.
231 students are taking LIVE classes
| Question Text | and 11.4.1 Preparation of Alcohols
Alcohols are prepared by the following methods:
1. From alkenes
(i) By acid catalysed hydration: Alkenes react with water in the presence of acid as catalyst to form alcohols. In case of unsymmetrical alkenes, the addition reaction takes place in accordance with Markovnikov's rule (Unit 13, Class XI).
Mechanism
The mechanism of the reaction involves the following three steps:
Step 1: Protonation of alkene to form carbocation by electrophilic attack of .
Step 2: Nucleophilic attack of water on carbocation.
|
| Updated On | May 28, 2023 |
| Topic | Alcohols, Phenols & Ethers |
| Subject | Chemistry |
| Class | Class 11 |
| Answer Type | Video solution: 1 |
| Upvotes | 123 |
| Avg. Video Duration | 5 min |