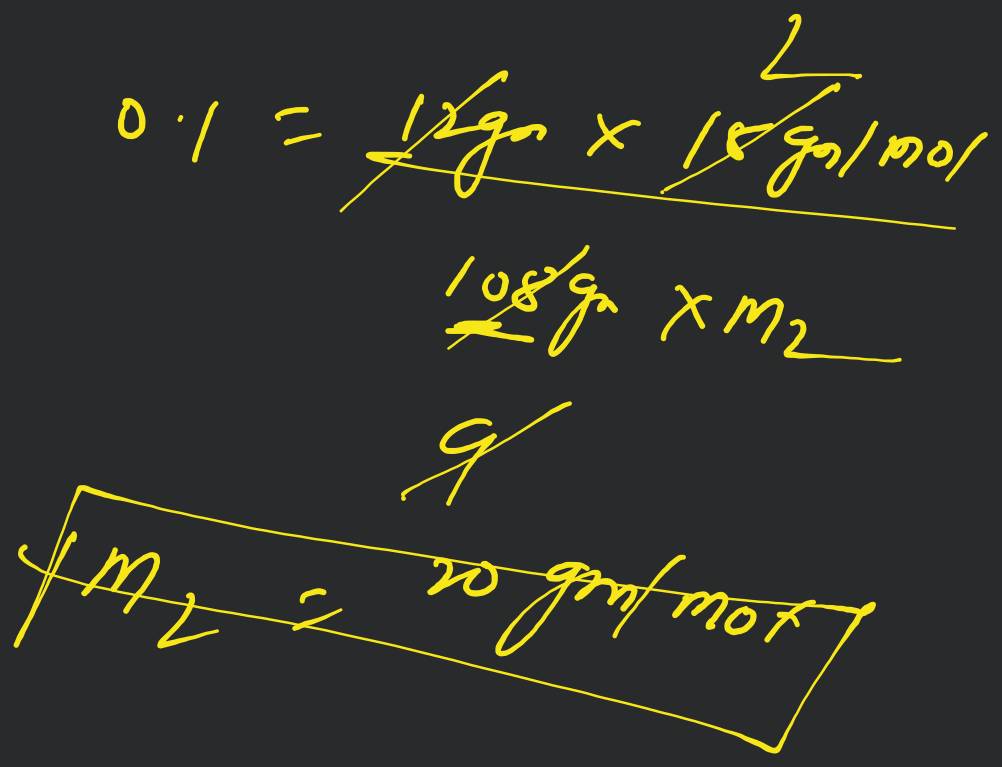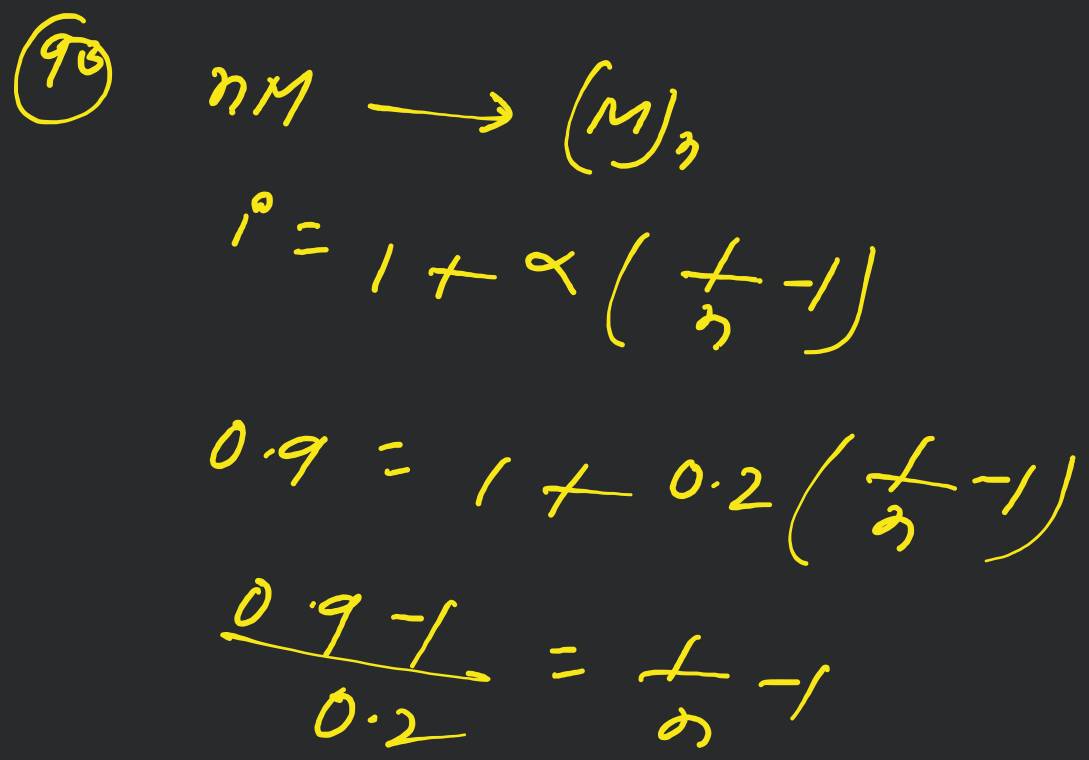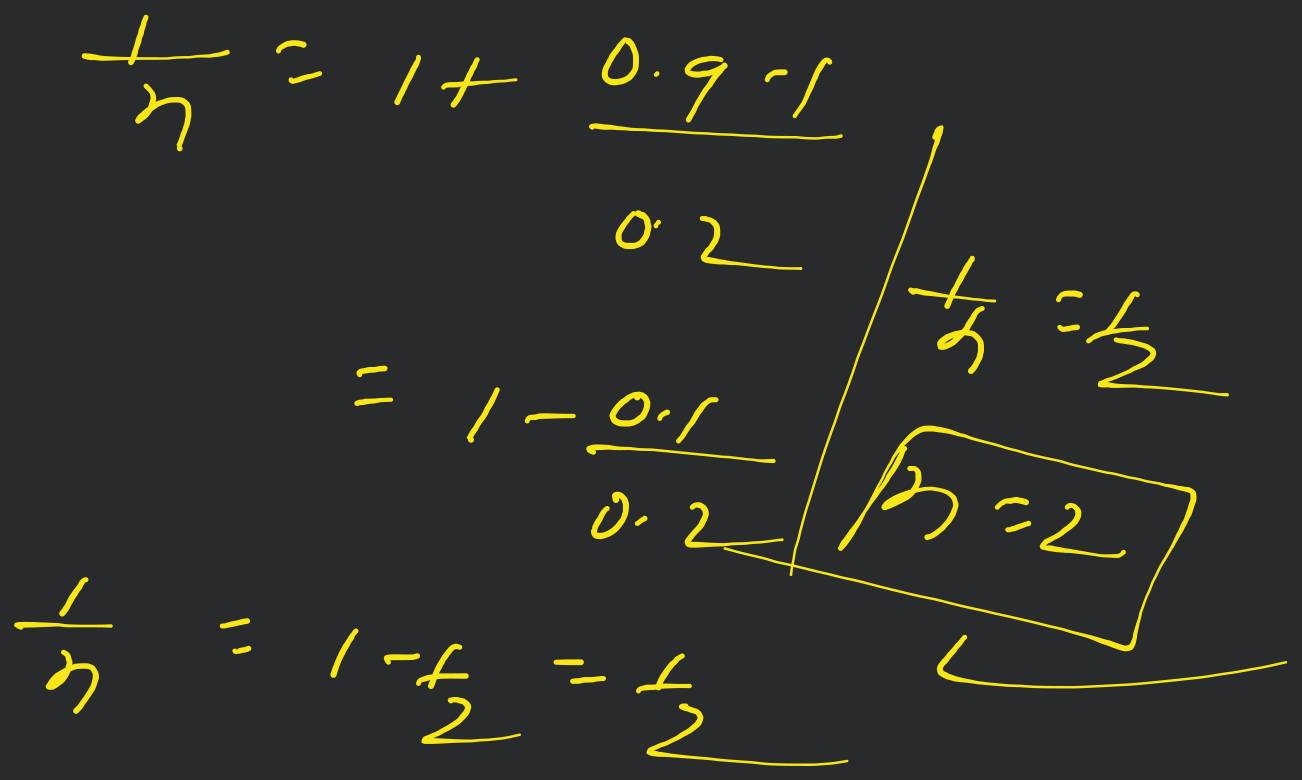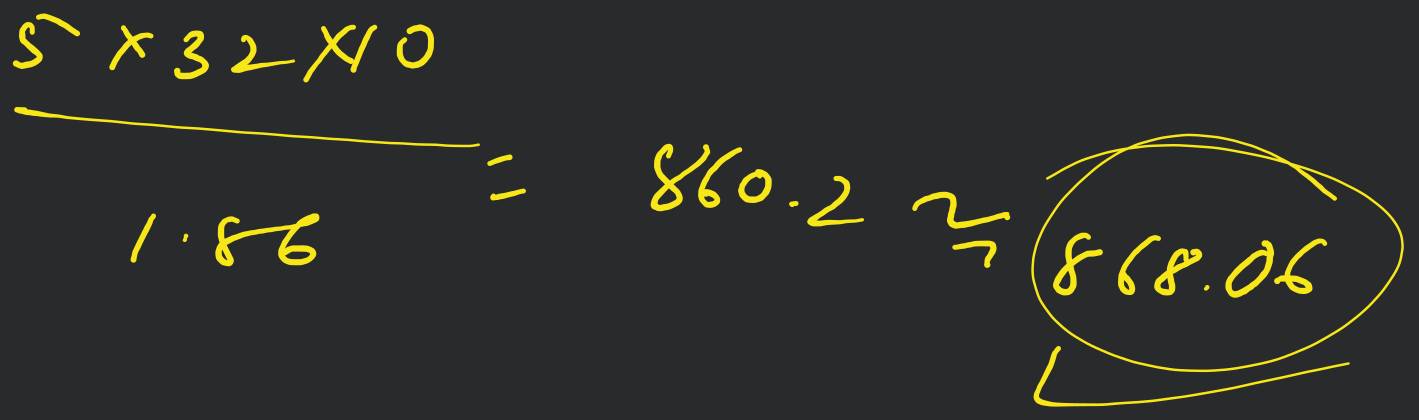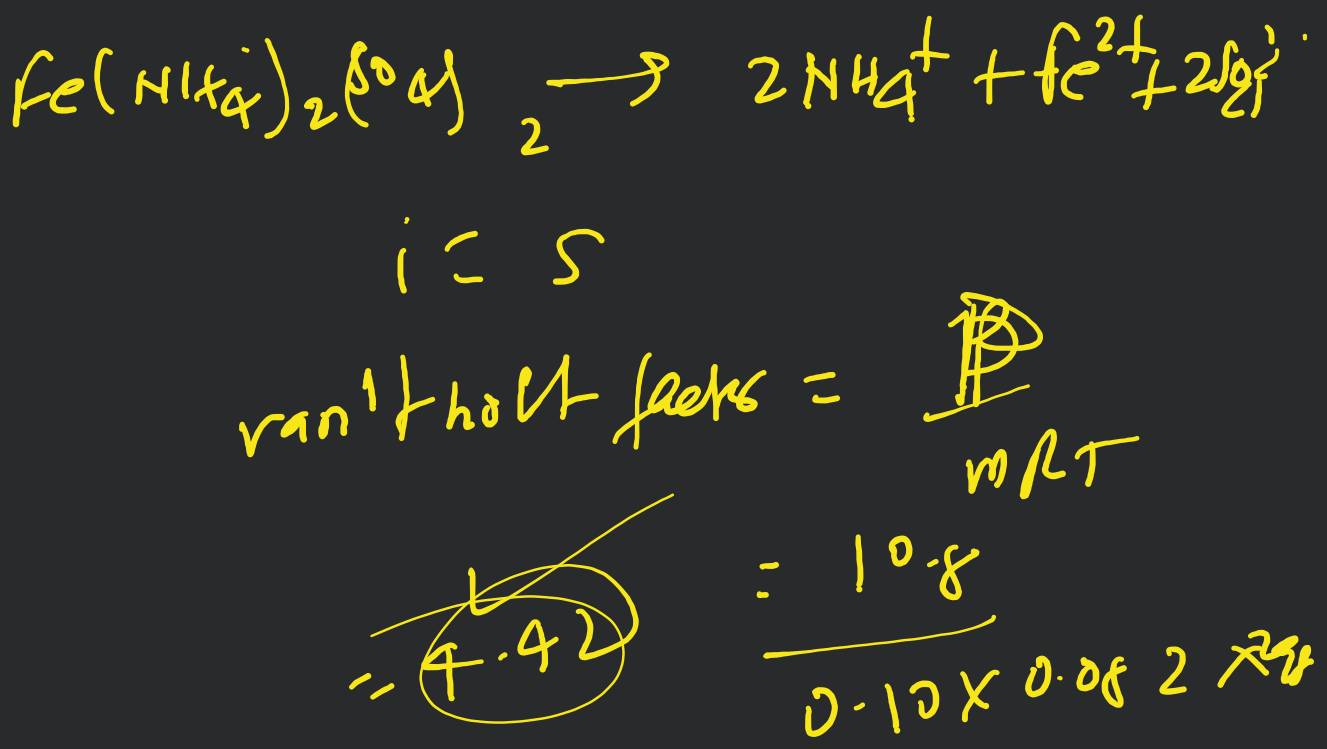Question
Question asked by Filo student
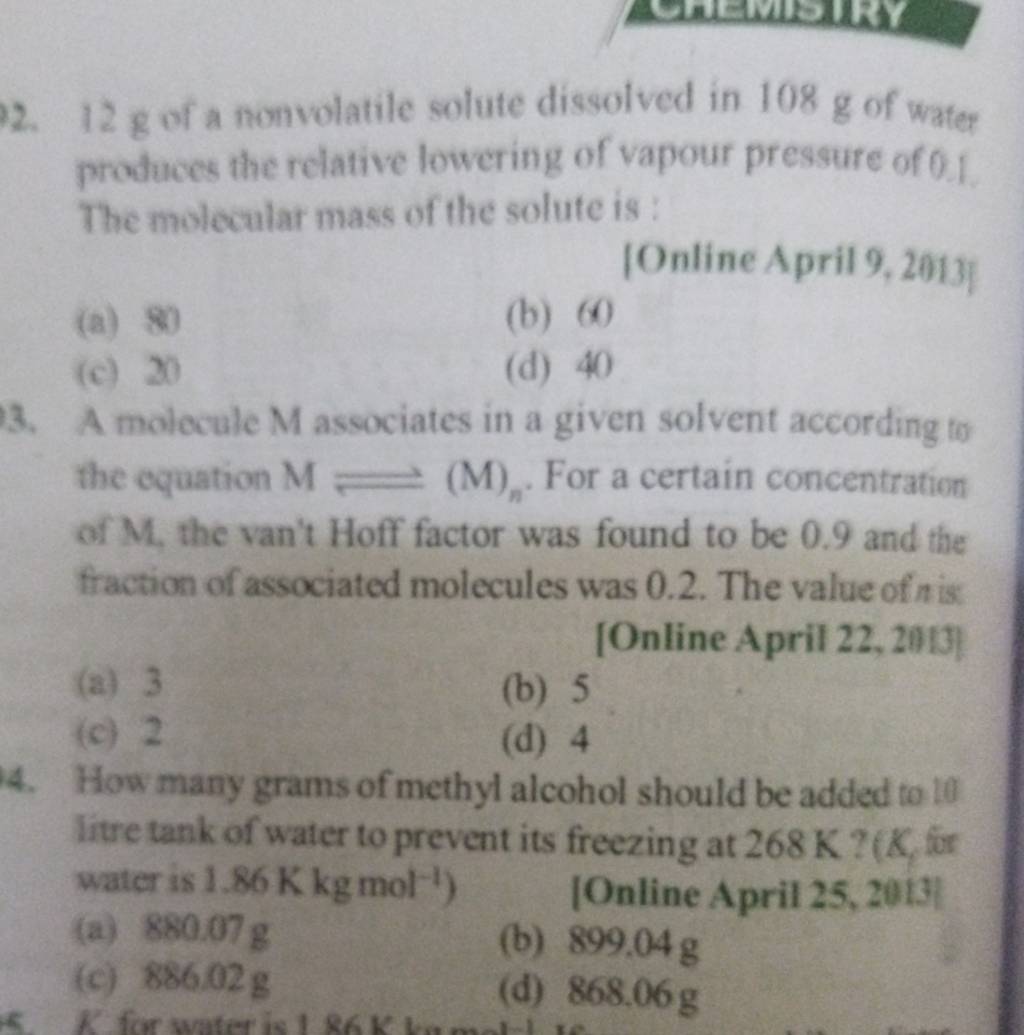
A molocule associates in a given solvent according to the equation . For a certain concentration of , the van't Hoff factor was found to be 0.9 and the fraction of associated molecules was 0.2 . The value of is [Online April 22, 20!1]
- 3
- 5
- 2
Found 7 tutors discussing this question
Discuss this question LIVE
14 mins ago

One destination to cover all your homework and assignment needs
Learn Practice Revision Succeed

Instant 1:1 help, 24x7
60, 000+ Expert tutors

Textbook solutions
Big idea maths, McGraw-Hill Education etc

Essay review
Get expert feedback on your essay

Schedule classes
High dosage tutoring from Dedicated 3 experts
Practice more questions on Gases & Solutions
Question 1
Easy
Views: 5,805
Question 2
Hard
Views: 5,817
Question 3
Views: 5,948
Question 4
Easy
Views: 5,250
Students who ask this question also asked
Question 1
Views: 5,925
Question 2
Views: 5,374
Question 3
Views: 5,580


Stuck on the question or explanation?
Connect with our Chemistry tutors online and get step by step solution of this question.
231 students are taking LIVE classes
| Question Text | A molocule associates in a given solvent according to the equation . For a certain concentration of , the van't Hoff factor was found to be 0.9 and the fraction of associated molecules was 0.2 . The value of is [Online April 22, 20!1] |
| Updated On | May 2, 2023 |
| Topic | Gases & Solutions |
| Subject | Chemistry |
| Class | Class 12 |
| Answer Type | Video solution: 1 |
| Upvotes | 139 |
| Avg. Video Duration | 14 min |