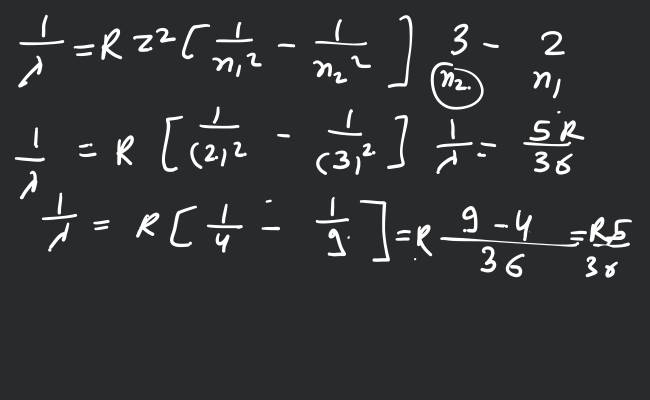Question
Medium
Solving time: 3 mins
In a hydrogen like atom, when an electron jumps from the -shell to the -shell, the wavelength of emitted radiation is . If an electron jumps from -shell to the -shell, the wavelength of emitted radiation will be
(a)
(b)
(c)
(d)
Found 7 tutors discussing this question
Discuss this question LIVE
12 mins ago
 Text solution
Text solution Verified
Verified
For hydrogen or hydrogen like atoms, we know that
where, is Rydberg constant and is atomic number. When electron jumps from - shell to the - shell, then
for
(for } M {-shell) } \therefore \left.\frac{1}{\lambda}=R Z^{2}\left[\frac{1}{2^{2}}-\frac{1}{3^{2}}\right]=\frac{5}{36} R Z^{2}\right]NLn_{1}=2 n_{2}=4 \therefore\frac{1}{\lambda^{\prime}}=R Z^{2}\left[\frac{1}{2^{2}}-\frac{1}{4^{2}}\right]=\frac{3}{16} R Z^{2}\frac{\lambda^{\prime}}{\lambda}=\left(\frac{5}{36} R Z^{2}\right) \div\left(\frac{3}{16} R Z^{2}\right)=\frac{20}{27}\lambda^{\prime}=\frac{20}{27} \lambda
where, is Rydberg constant and is atomic number. When electron jumps from - shell to the - shell, then
for
(for } M {-shell) } \therefore \left.\frac{1}{\lambda}=R Z^{2}\left[\frac{1}{2^{2}}-\frac{1}{3^{2}}\right]=\frac{5}{36} R Z^{2}\right]NLn_{1}=2 n_{2}=4 \therefore\frac{1}{\lambda^{\prime}}=R Z^{2}\left[\frac{1}{2^{2}}-\frac{1}{4^{2}}\right]=\frac{3}{16} R Z^{2}\frac{\lambda^{\prime}}{\lambda}=\left(\frac{5}{36} R Z^{2}\right) \div\left(\frac{3}{16} R Z^{2}\right)=\frac{20}{27}\lambda^{\prime}=\frac{20}{27} \lambda
Was this solution helpful?
27
Share
Report
Filo tutor solutions (2)
Learn from their 1-to-1 discussion with Filo tutors.

One destination to cover all your homework and assignment needs
Learn Practice Revision Succeed

Instant 1:1 help, 24x7
60, 000+ Expert tutors

Textbook solutions
Big idea maths, McGraw-Hill Education etc

Essay review
Get expert feedback on your essay

Schedule classes
High dosage tutoring from Dedicated 3 experts
Questions from JEE Mains 2013 - PYQs
Question 1
Medium
Views: 5,522
(a)
(b)
(c)
(d)
Question 2
Medium
Views: 5,237
(a)
(b)
(c)
(d)
Question 3
Easy
Views: 5,622
Statement I: Higher the range, greater is the resistance of ammeter.
Statement II: To increase the range of ammeter, additional shunt needs to be used across it.
(a) Statement I is true, Statement II is true; Statement II is the correct explanation of Statement I
(b) Statement I is true, Statement II is true; Statement II is not the correct explanation of Statement I
(c) Statement I is true, Statement II is false
(d) Statement I is false, Statement II is true
Question 4
Medium
Views: 5,698
(a)
(b)
(c)
(d)
Practice questions from Arihant Physics JEE Main Chapterwise Solutions (2019-2002) (Arihant)
Question 1
Medium
Views: 5,867
(a)
(b)
(c)
(d)
Question 2
Medium
Views: 5,888
Question 3
Medium
Views: 5,937
(a)
(b)
(c)
(d)
Question 4
Medium
Views: 5,256
Practice questions from Atoms in the same exam
Question 1
Medium
Views: 5,598
The value of x, to the nearest integer, is
Question 2
Medium
Views: 5,472
(a)
(b)
(c)
(d)
Practice more questions from Atoms
Question 1
Medium
Views: 6,357
Question 2
Medium
Views: 5,330
(a) Find the ionization potential of this atom.
(b) Find the short wavelength limit of the series terminating at n = 2.
(c) Find the excitation potential for the state n = 3.
(d) Find wave number of the photon emitted for the transition n = 3 to n = 1.
Question 4
Hard
Views: 6,093
(a) radius of third orbit for ,
(b) speed of electron in fourth orbit for .
Practice questions on similar concepts asked by Filo students
Question 1
Views: 5,981
Question 2
Views: 5,624
Question 3
Views: 5,582
Question 4
Views: 5,175


Stuck on the question or explanation?
Connect with our Physics tutors online and get step by step solution of this question.
231 students are taking LIVE classes
| Question Text | In a hydrogen like atom, when an electron jumps from the -shell to the -shell, the wavelength of emitted radiation is . If an electron jumps from -shell to the -shell, the wavelength of emitted radiation will be (a) (b) (c) (d) |
| Updated On | Feb 28, 2023 |
| Topic | Atoms |
| Subject | Physics |
| Class | Class 12 |
| Answer Type | Text solution:1 Video solution: 2 |
| Upvotes | 234 |
| Avg. Video Duration | 8 min |