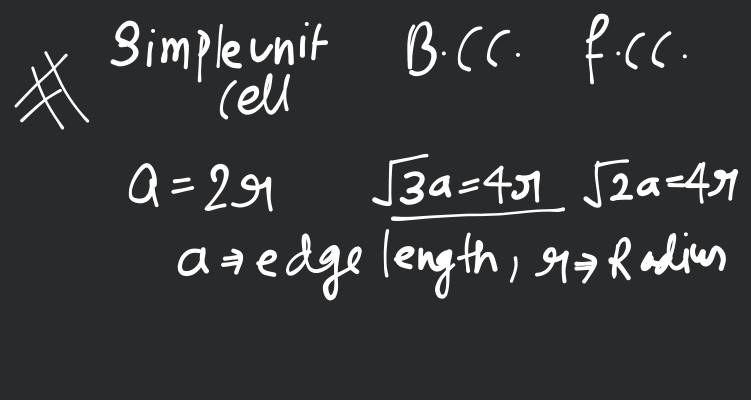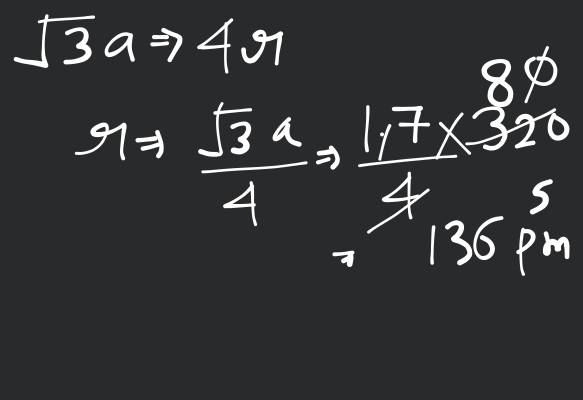Question
Medium
Solving time: 3 mins
Sodium metal crystallises in a body centred cubic lattice with a unit cell edge of . The radius of sodium atom is approximately
Found 3 tutors discussing this question
Discuss this question LIVE
13 mins ago
 Text solution
Text solution Verified
Verified
From this figure ,
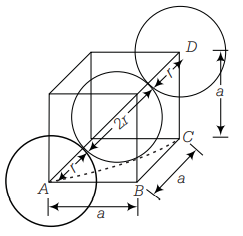 Now, when Na metal crystallises in bcc unit cell with unit cell edge,
We have the formula for radius, i.e.
Now, when Na metal crystallises in bcc unit cell with unit cell edge,
We have the formula for radius, i.e.
Was this solution helpful?
51
Share
Report
Filo tutor solutions (3)
Learn from their 1-to-1 discussion with Filo tutors.
20 mins
Uploaded on: 2/24/2023
Was this solution helpful?
76
Share
Report
2 mins
Uploaded on: 5/11/2023
Was this solution helpful?
150
Share
Report

One destination to cover all your homework and assignment needs
Learn Practice Revision Succeed

Instant 1:1 help, 24x7
60, 000+ Expert tutors

Textbook solutions
Big idea maths, McGraw-Hill Education etc

Essay review
Get expert feedback on your essay

Schedule classes
High dosage tutoring from Dedicated 3 experts
Questions from JEE Mains 2015 - PYQs
Question 2
Medium
Views: 5,912
Question 4
Medium
Views: 6,335
Practice questions from Arihant Chemistry JEE Main Chapterwise Solutions (2019-2002) (Arihant)
Question 1
Easy
Views: 5,697
Question 2
Medium
Views: 6,028
Question 3
Easy
Views: 5,729
Question 4
Medium
Views: 5,965
Practice questions from The Solid State in the same exam
Question 1
Easy
Views: 5,674
(A) Crystalline solids have long range order.
(B) Crystalline solids are isotropic
(C) Amorphous solids are sometimes called pseudo solids
(D) Amorphous solids soften over a range of temperature
(E) Amorphous solids have a definite heat of fusion
Choose the most appropriate answer from the options given below:
Question 2
Hard
Views: 5,713
[Given : ]
Question 3
Medium
Views: 5,823
Assertion A : Sharp glass becomes smooth on heating it up to its melting point.
Reason R : The viscosity of glass decreases on melting.
Choose the most appropriate answer from the options given below :
Practice more questions from The Solid State
Question 1
Easy
Views: 5,721
Question 3
Easy
Views: 5,876
Practice questions on similar concepts asked by Filo students
Question 1
Views: 5,995
Question 4
Views: 5,101


Stuck on the question or explanation?
Connect with our Chemistry tutors online and get step by step solution of this question.
231 students are taking LIVE classes
| Question Text | Sodium metal crystallises in a body centred cubic lattice with a unit cell edge of . The radius of sodium atom is approximately |
| Updated On | May 11, 2023 |
| Topic | The Solid State |
| Subject | Chemistry |
| Class | Class 12 |
| Answer Type | Text solution:1 Video solution: 3 |
| Upvotes | 416 |
| Avg. Video Duration | 8 min |