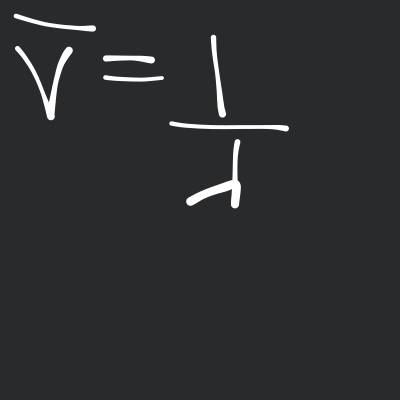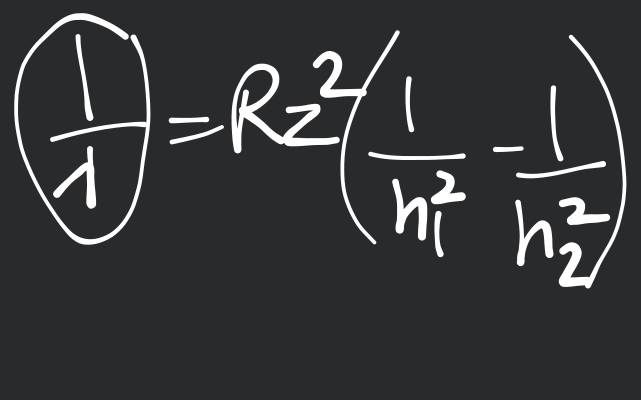Question
Easy
Solving time: 2 mins
Hydrogen atom is excited from ground state to another state with principal quantum number equal to 4 . Then, the number of spectral lines in the emission spectra will be
(a) 2
(b) 3
(c) 5
(d) 6
Found 5 tutors discussing this question
Discuss this question LIVE
15 mins ago
 Text solution
Text solution Verified
Verified
In emission spectrum, number of bright lines is given by
Was this solution helpful?
76
Share
Report

One destination to cover all your homework and assignment needs
Learn Practice Revision Succeed

Instant 1:1 help, 24x7
60, 000+ Expert tutors

Textbook solutions
Big idea maths, McGraw-Hill Education etc

Essay review
Get expert feedback on your essay

Schedule classes
High dosage tutoring from Dedicated 3 experts
Questions from JEE Mains 2013 - PYQs
Question 1
Easy
Views: 5,622
Statement I: Higher the range, greater is the resistance of ammeter.
Statement II: To increase the range of ammeter, additional shunt needs to be used across it.
(a) Statement I is true, Statement II is true; Statement II is the correct explanation of Statement I
(b) Statement I is true, Statement II is true; Statement II is not the correct explanation of Statement I
(c) Statement I is true, Statement II is false
(d) Statement I is false, Statement II is true
Question 2
Medium
Views: 5,498
(a)
(b)
(c)
(d)
Question 3
Medium
Views: 5,772
Question 4
Medium
Views: 5,513
Practice questions from Arihant Physics JEE Main Chapterwise Solutions (2019-2002) (Arihant)
Question 1
Medium
Views: 5,350
Question 2
Easy
Views: 5,821
(a) Heisenberg's uncertainty principle
(b) Pauli's exclusion principle
(c) Bohr's correspondence principle
(d) Boltzmann's law
Question 3
Medium
Views: 5,867
(a)
(b)
(c)
(d)
Question 4
Hard
Views: 5,523
(a)
(b)
(c)
(d)
Practice questions from Atoms in the same exam
Question 1
Easy
Views: 5,729
Question 2
Easy
Views: 6,091
Question 3
Easy
Views: 5,738
Practice more questions from Atoms
Question 1
Easy
Views: 5,821
Question 2
Medium
Views: 5,230
Question 3
Easy
Views: 5,136
and is the distance of the electron from the origin. By applying Bohr model to this
system, the radius of the th orbital of the electron is found to be and the kinetic
energy of the electron to be . Then which of the following is true?
Question 4
Hard
Views: 5,733
Practice questions on similar concepts asked by Filo students
Question 1
Views: 5,109
Question 2
Views: 5,732
Question 3
Views: 5,531
Question 4
Views: 5,350


Stuck on the question or explanation?
Connect with our Physics tutors online and get step by step solution of this question.
231 students are taking LIVE classes
| Question Text | Hydrogen atom is excited from ground state to another state with principal quantum number equal to 4 . Then, the number of spectral lines in the emission spectra will be (a) 2 (b) 3 (c) 5 (d) 6 |
| Updated On | Jan 30, 2023 |
| Topic | Atoms |
| Subject | Physics |
| Class | Class 12 |
| Answer Type | Text solution:1 Video solution: 1 |
| Upvotes | 215 |
| Avg. Video Duration | 12 min |